The development and application of resonance ionization (RI) with tunable lasers and its combination with mass spectrometric (MS) techniques opens up a broad field of research.
RIMS enables atomic-physical and quantum-optical investigations for basic research, the selective production of exotic ions at large-scale research facilities such as CERN in Geneva, and the analytical ultratrace determination of the rarest natural or anthropogenic species. In all these cases, the highest selectivity is required with regard to the suppression of interfering contamination of neighboring elements or isotopes. The ambitious specifications require continuous research and development both on the side of the laser systems used and on the mass spectrometric techniques of selecting, cooling, trapping and detecting the laser ions.
Due to the uniqueness of the spectral fingerprint of an element, resonant optical excitation of atoms ensures highest selectivity in the ionization of an element and completely suppresses neighboring elements (isobars). Spectrally narrowband continuous laser radiation can also ensure highest isotope selectivity by exploiting the isotope shift. The subsequent mass separation and single ion detection suppresses unspecific background and is a prerequisite for high efficiency and lowest detection limits. The specifications of the Mainz LARISSA (LAser Resonance Ionization for Spectroscopy in Selective Applications) exceed conventional techniques by far.
The cornerstone for the development and continuous improvement of RIMS technology is on the one hand the design and construction of the laser light sources and on the other hand the adaptation and optimization of the systems for mass spectrometry and ion manipulation. In addition, extensive spectroscopic investigations of the optical excitation and ionization paths of the respective element to be studied are required.
Principle
For the investigation of rarest isotopes, the isotopes of interest must be separated with high selectivity from the up to twenty orders of magnitude more common isotopes and other elements in the sample of interest. For this purpose we use the method of resonance ionization mass spectrometry, RIMS for short. This can be divided into three steps. These are the atomization of the sample, the resonant selective ionization and the subsequent mass separation of the ions.
Step 1: Atomization
During atomization, the sample to be analyzed is placed in a furnace. At temperatures up to 2000 K the sample is evaporated and most moleculular species are thermally broken up. Due to the geometry of the furnace, a collimated atomic beam is created.
Step 2: Resonance Ionization
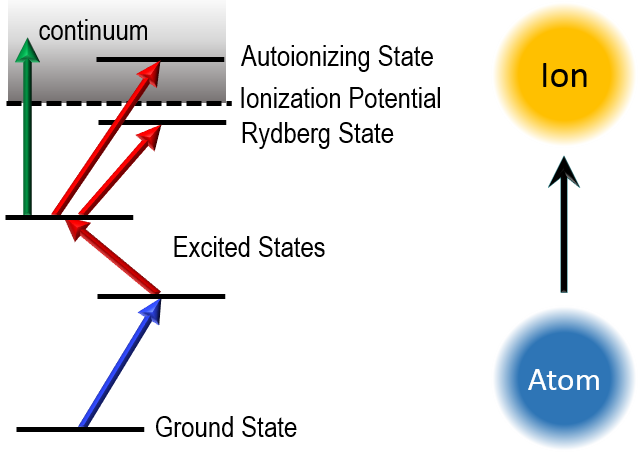
The most distinctive atomic property of an element are the energy levels of the atoms. If only one element is to be excited selectively, a laser is tuned in such a way that the energy transfer corresponds exactly to the difference between two characteristic energy levels. The narrower the energy distribution, i.e. the narrower the spectral linewidth, the more selectively an atom can be excited.
If the spectral energy distribution in the individual excitation steps is narrower than the isotopic shift of the isotopes of an element, even a monoisotopic ion beam can be prepared from the sample. Ions generated in this process are often called laser ions.
 |
| Schematic of Resonance Ionization |
Step 3: Mass separation
To enable an even sharper selection and to distinguish the laser ions from other ions, specifically an unwanted isotope, the ions are focused by ion optics into a mass filter through which only ions of a single mass are transmitted. These re-sorted ions are then detected and counted on a detector.
Implementation
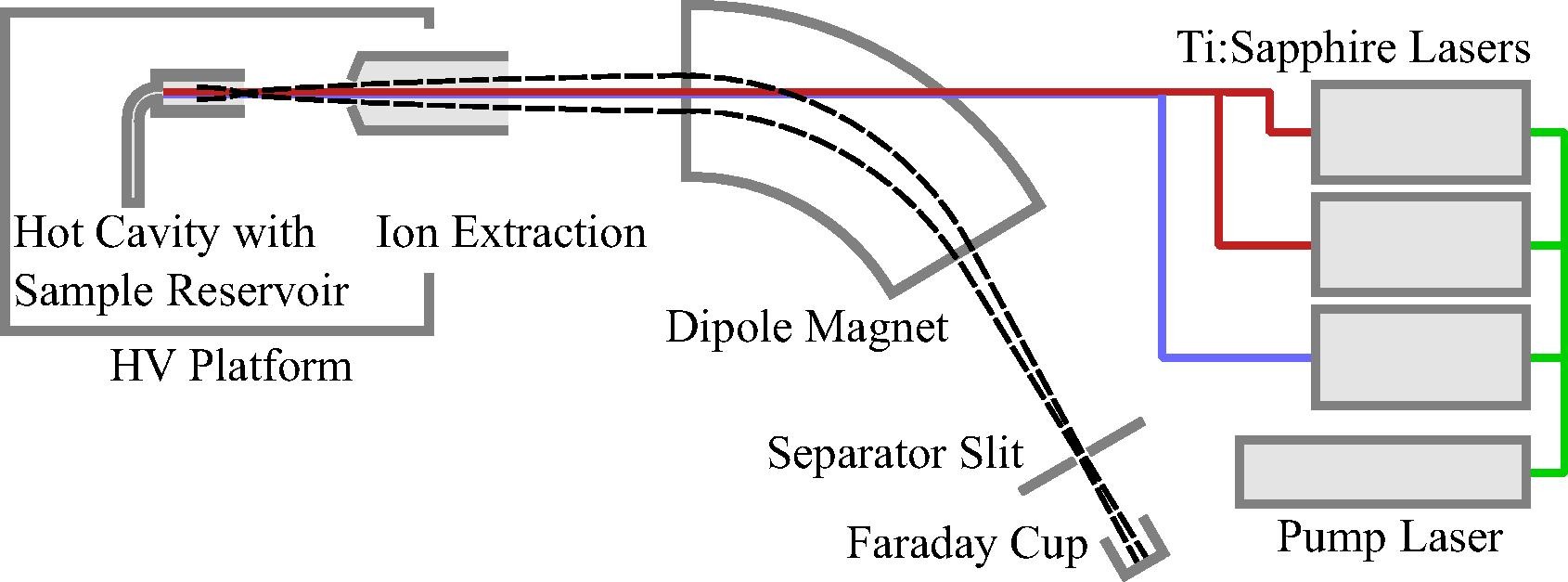
The RISIKO Mass separator
The sample is usually introduced as an acidic solution into a graphite or tantalum furnace and heated electrothermally at up to 2000 K. The collimated atomic beam is then overlapped with the laser light of three lasers. For selective excitation of the atoms we use self-constructed titanium:sapphire lasers. Optionally and depending on the element, the sought-after isotopes are excited into an autoionizing state or a high Rydberg state, which further increases the ionization efficiency due to the fully resonant ionization process.
 |
| In the LARISSA Group Ti:Sapphire lasers are specially developed and tailored to RIMS applications |
The laser ions are directed into a dipole magnet by optimized ion optics and separated according to their mass-to-charge ratio. The transmitted ions are detected by a secondary electron multiplier or a Faraday cup.
The spectral detuning and stabilization of the lasers, the control of the mass separator as well as the data acquisition and display are performed by a self-developed software on a standard PC.